 The IOM, P2P and AHRQ reports all pointed out a serious gap in ME/CFS research: the absence of validated ways of assessing clinical outcomes. I have new information about an initiative to change that, and I’ll be speaking about my experiences as an ME/CFS patient representative at an FDA public meeting tomorrow.
The IOM, P2P and AHRQ reports all pointed out a serious gap in ME/CFS research: the absence of validated ways of assessing clinical outcomes. I have new information about an initiative to change that, and I’ll be speaking about my experiences as an ME/CFS patient representative at an FDA public meeting tomorrow.
How do you know if a treatment is working? How do you know if your disease is getting better or worse? As an individual, this is something to discuss with your healthcare provider. But in the context of a research study or a clinical trial, we need ways to assess this systematically. One way to do this is with a clinical outcomes assessment tool.
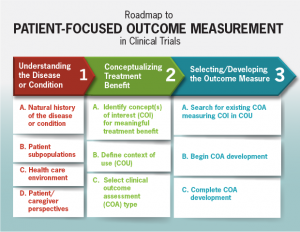
Clinical outcomes assessment (or COA) measures changes in how a patient feels or functions, and can be based on patient report, clinician assessment, or similar measures. It is different from a biomarker, which is an objective physical measurement. A COA can be a questionnaire or a measure of performance, and it has to measure something that matters (like a symptom or ability to function). This is really important for FDA because there has to be a way to prove a treatment is working.
But you can’t just pick a random set of questions and start using it, and expect FDA to accept it. FDA has a whole process of COA qualification. And it can be expensive to do the necessary research to prove that your questionnaire measures what you want in the patients who will use it.
After the April 2013 FDA meeting on Patient Focused Drug Development in ME/CFS, FDA took steps to address the need for a qualified COA in ME/CFS. FDA convened an ME/CFS Outcomes Measures Working Group comprised of representatives from FDA, NIH, CDC, and academia. Last year, I joined this group in the capacity of patient representative. While FDA is not developing the COA itself, the Working Group is collaborating on a proposal for funding to conduct the necessary research.
While I can’t share specifics about the proposal at this point in time, I can share my impressions from working with the group. First of all, I think it is fair to say that FDA is highly motivated to make progress on ME/CFS. The agency is not developing the COA tool itself, but they really want to help make it happen. Second, I have been able to participate as an equal member in the Working Group and in sidebar conversations. I have been treated as an equal, and my perspective as a patient has been sought out by the proposal team. Even better, my experiences as a patient have had an impact on the group’s work. This is a group that is open to data and information, and opinions shift as a result.
I have not often had the experience of feeling heard by government employees. This is why I applied for the FDA’s Patient Representative program in the first place: to make sure FDA hears the perspectives of ME/CFS patients whenever possible. It has been rewarding to share my experiences with a group actively working to create a COA for use in ME/CFS research and drug development.
If you would like to learn more about COAs and how FDA plans to incorporate patient voices in the process, you can watch an all-day public meeting on Wednesday, April 1, 2015. I have been invited to participate in the last panel of the meeting on the patient perspective on how to move forward. I’m not able to travel to Maryland for the meeting, so will be speaking by webcast.

Jennie, wow–you are working really hard for us, and being heard–we owe you a lot! Don’t wear out–we need you long-term! But thanks for sharing some good news about what CDC is doing–sounds as if real change is coming!
Chris
Jennie, I’m relatively new to this site, but I’m not new to this sickness, I been sick for 25 years, last 5 years it got really bad and if it was not for people like you we would be all a forgotten nation. Thank you, thank you, thank you. You are my hero !
Thank you, Sofia. There are a lot of advocates doing good work. I was fortunate to land in the FDA program at the right time.
While I remain skeptical of clinical outcomes assessment, I am delighted that government reps are actually listening to what you have to say. This is a new sensation; please bottle and sell it.
Today’s million dollar idea – bottled hope and progress!
“If you would like to learn more about COAs and how FDA plans to incorporate patient voices in the process, you can watch an all-day public meeting on Wednesday, April 1, 2015.”
This isn’t April Fools is it ? 😉
All this news –Columbia publications, Strong Advocate meeting in DC, and now FDA wants to help — coming out lately is getting to me.
Once again, thank you for your help on behalf of us all Jennie.
It’s the real deal, Cheryl! Thank you for your kind words.
Jennie,
I signed up for the FDA webcast and hopefully will be able to listen to some of it in between traveling to a doctors appointment. I was thrilled to see you were on the agenda! I just want to say thanks for your continued advocacy efforts. Your input has been invaluable and I, for one, really appreciate it.
Jennie, I only just now learned of this webcast, so I was unable to register for it. Any chance it will be posted on YouTube? If I’m not mistaken, you are the same Jennie who has had your letter read on TWiV lately, are you not? Thank you for all you are doing. I WISH I was still coherent enough to take on discussions of this nature. As it is, my conversations are mostly ‘fill-in-the-blank’ and sound rather unintelligent. God bless you! Keep them all on track!
Yes, that was my letter on TWiV! The blog post below this one has both of my letters. FDA usually posts their public meeting videos online, and I’ll try to remember to post the link. Thank you!
Thanks for this report, Jennie. I’m late getting to my emails and missed the FDA meeting that you mention was going to take place today. Is there some way I can watch a replay? Can you give us the link? Thanks. Keep up your wonderful, valuable participation on ME patients’ behalf. 🙂
The FDA really wants to do something useful? I’m glad I was lying down to read this.
Now, how do we pry loose some research money from NIH? Throw a retirement party for Dr Fauci? He’s been at NIH since the Nixon era – he must’ve earned a few years off by now…
I’m looking for places to get good images of easy software to use to make images. I wondered if you’d mind sharing how you made the image in this blog and other blogs.
Maybe you could email me back rather than cluttering up your post with unrelated questions. I just wasn’t sure how else to contact you. Thanks
I imagine the image is copied from this page:
http://www.fda.gov/Drugs/DevelopmentApprovalProcess/DrugDevelopmentToolsQualificationProgram/ucm284077.htm
Yes, I used an FDA slide for the image.
Jennie, where in the FDA meeting are you (minutes as shown on video) ? I have heard good things about your talk – no surprise there! – and also the one of the fellow from PROMIS. I can’t listen to the whole day’s worth of meeting. Any other parts that are really good? If so, please give us the times that are relevant.
A far less favorable aspect of the FDA involves their drug approval process. Better than nothing that they are listening to patients about validated outcome methods and having all-day seminars on them. However, there’s been a drug in the FDA pipeline for 20 years without an approval! While the FDA does at this time have the regulatory paths to allow for conditional approval of Ampligen, it refuses to use these paths. Which is more important to most patients – a process for creating validated ways of assessing clinical outcomes or a drug approval that might help them feel much better? Those who have been on Ampligen for many years as part of the trial have been given at least some of their lives back. This drug should be available to all ME/SEID patients. Plus, one drug approved for this disease will bring in more pharmaceutical companies for approval, some, one would hope which already have drugs being used off-label to treat ME/SEID.
Once the video is archived, I’ll post the times of my comments.
That’s great news Jennie – thanks for sharing it 🙂
Thank you for sharing this information, it really made my day! I didn’t read this in time to watch, but look forward to seeing it if it gets online somewhere. Thank you for being an advocate for all of us patients that can’t think straight enough to participate in anything that taxing! 🙂 Here’s hoping for a brighter future for all of us
Thank you for sharing this information, it really made my day! I didn’t read this in time to watch, but look forward to seeing it if it gets online somewhere. Thank you for being an advocate for all of us patients that can’t think straight enough to participate in anything that taxing! 🙂 Here’s hoping for a brighter future for all of us